Sensori per pulsossimetria
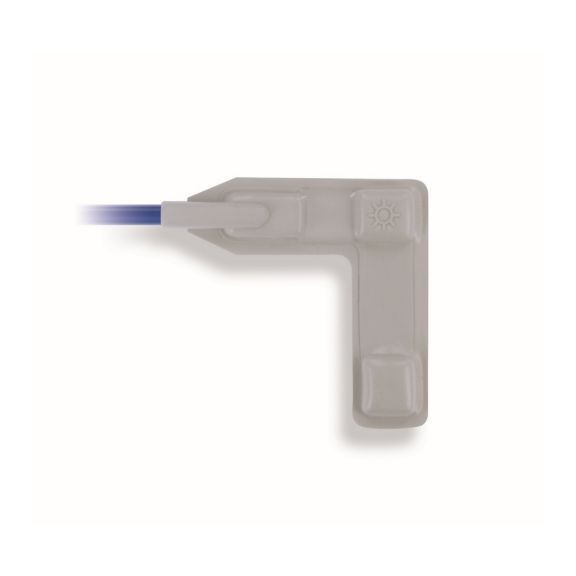
Wrap Sensors
Reusable wrap pulse oximetry sensors for neonates to adults. Flexible application. Easy to clean
Please sign in to access more documents
Once signed in, you may be able to access additional documents for your account.
EnviteC Cross Reference List | Tech Support
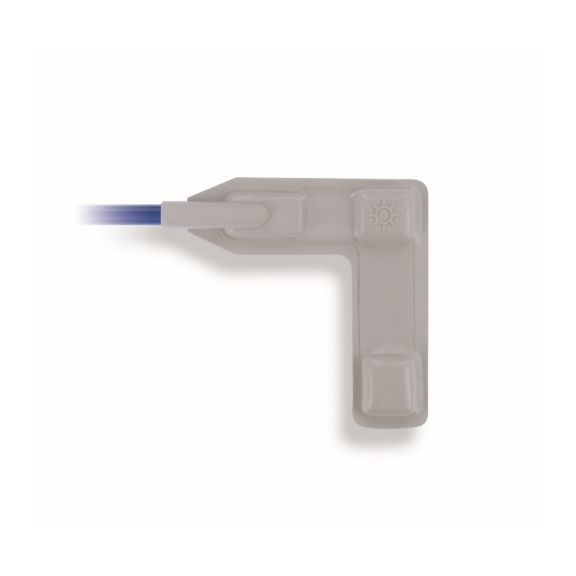
Reusable wrap pulse oximetry sensors that wrap. Easy to clean and offer flexible applications
EnviteC by Honeywell
Certificato
Altro
Per favore accedi per visualizzare i codici dei componenti disponibili per l'acquisto in base al tuo account Registrazione
Per favore accedi per visualizzare i codici dei componenti disponibili per l'acquisto in base al tuo account Registrazione