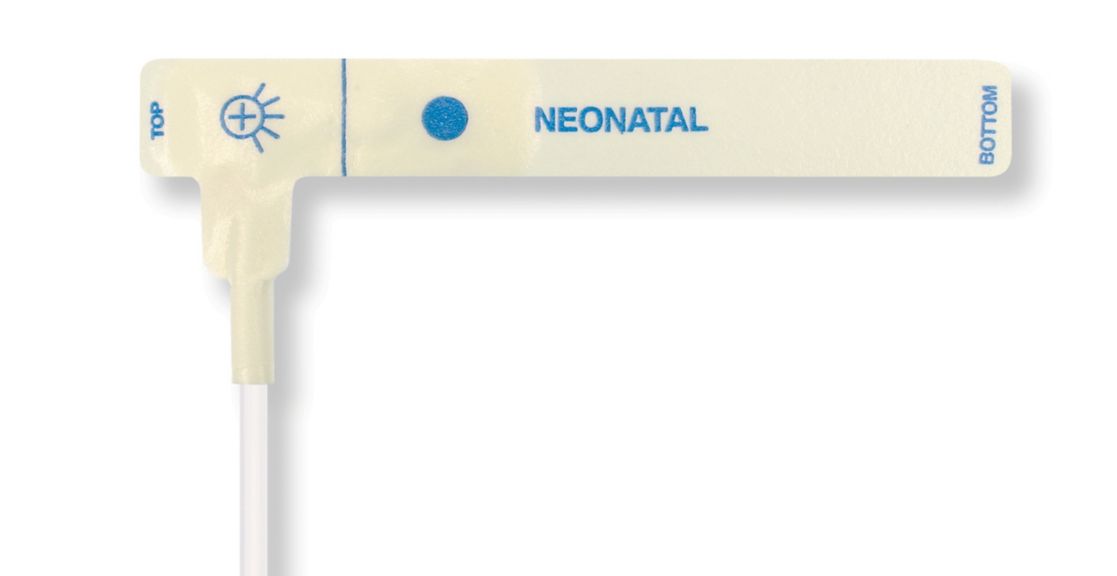
Pulse Oximetry Sensors
Disposable Pulse Oximetry Sensors for Neonates
• Good fit • Flexible and adaptable • Easy to use
Please sign in to access more documents
Once signed in, you may be able to access additional documents for your account.
Certificate
Others
Instructions for Use
Please sign in to view part numbers available for purchase based on your account Sign In
Please sign in to view part numbers available for purchase based on your account Sign In