pulse oximetry sensors
パルスオキシメータ&アクセサリ
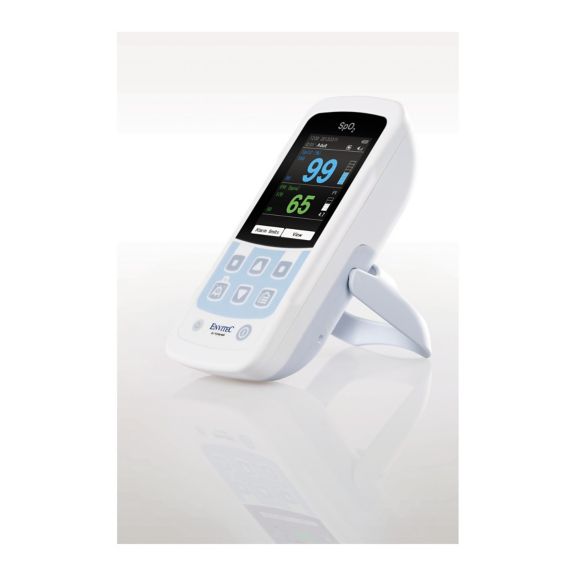
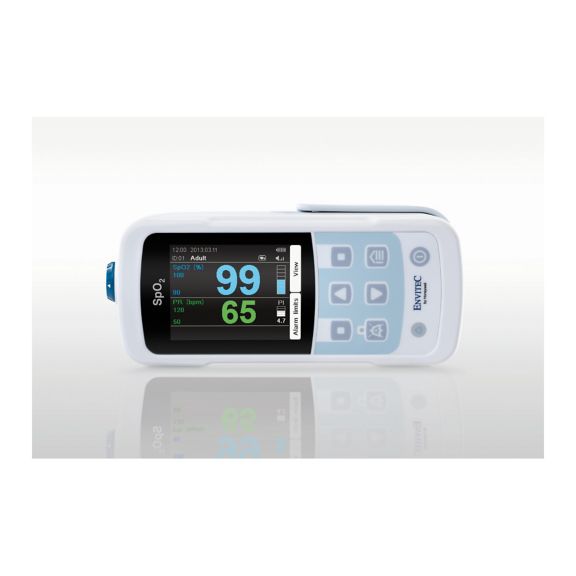
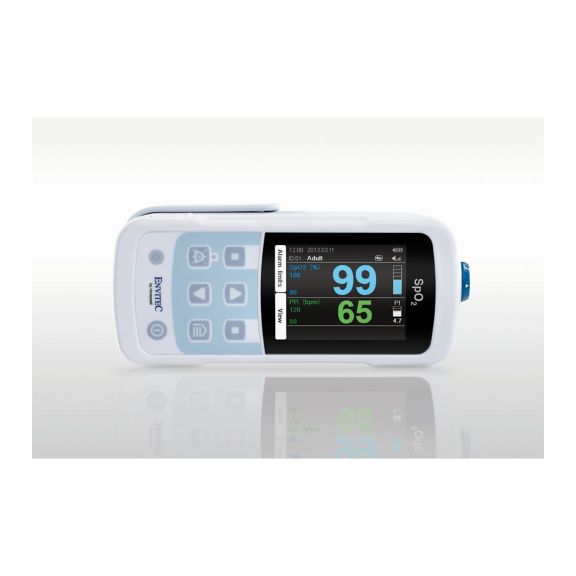
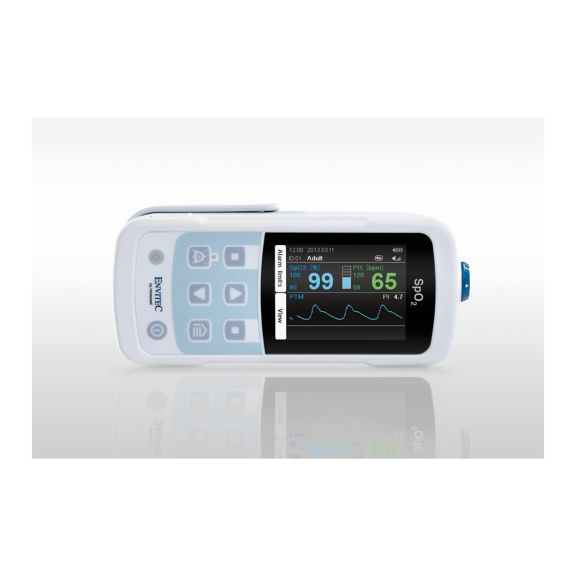
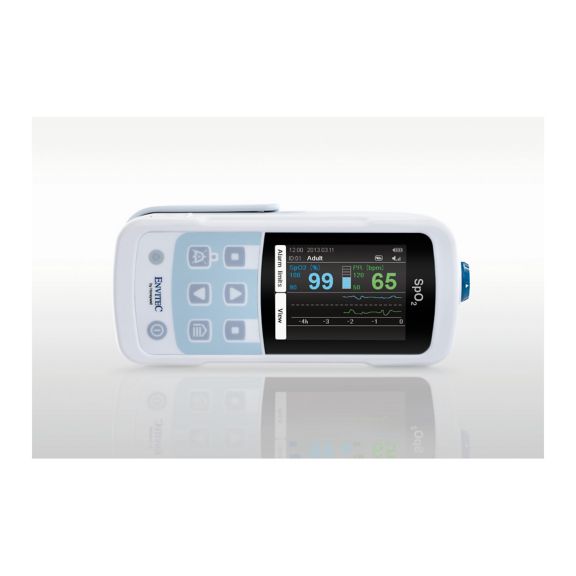
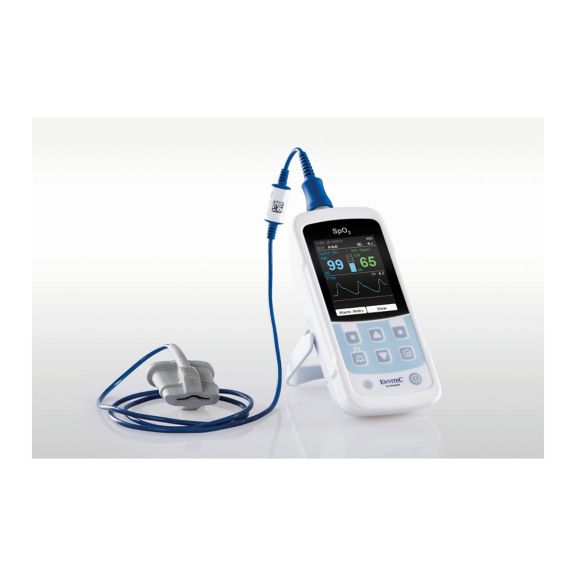
さまざまなオプションが可能なパルスオキシメータです。連続およびスポット測定のSpO2モニタリングに適しています。ポータブルまたは据え置き型のデバイスとして設計されています。SpO2、脈拍数を測定し、プレチスモグラフによる脈波を表示します。
Please sign in to access more documents
Once signed in, you may be able to access additional documents for your account.
パルスオキシメータは、動脈血酸素飽和度(SpO2)と脈拍数を測定する、非侵襲的でシンプルかつ信頼性の高い技術です。
小型で携帯可能なハンディ型パルスオキシメータモニターは、医療現場でさまざまな用途で使用されています。SpO2センサは、医療行為には不可欠なものです。患者さんの装着部位はさまざまです。
MySign® S は、高精度な測定機能と使いやすいモニターを備えた、バイタルサインを非侵襲的にモニタリングするためのインテリジェントなソリューションです。
ハネウェル製EnviteC
より多くのドキュメントにアクセスするためには、サインインをしてください。
サインインすることで追加ドキュメントにアクセスできるようになります。
ブローシャ―
認証
その他
お願いします サインインすると、アカウントに基づいて購入可能な部品番号が表示されます サインイン