pulse oximetry sensors
Wrap
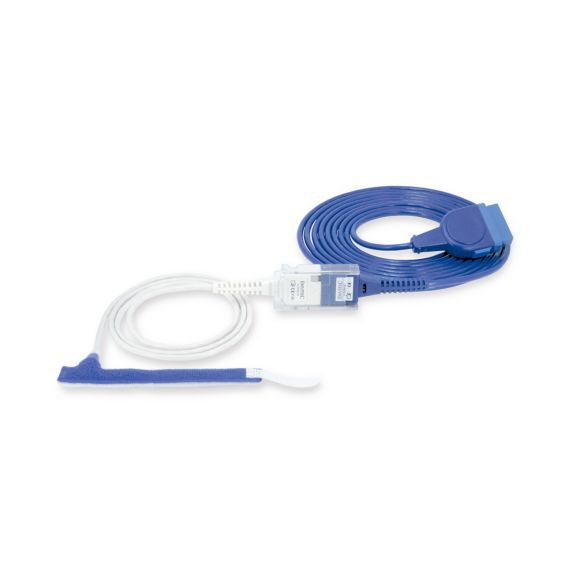
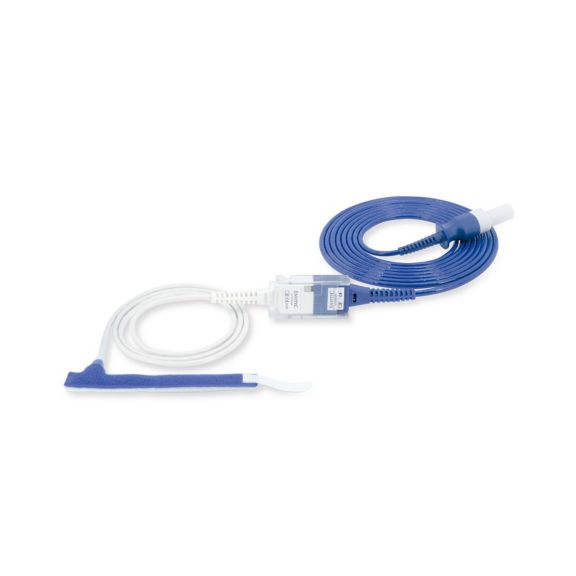
Reuseable wrap pulse oximetry sensors for neonates to adults. Flexible application. Easy to clean
Please sign in to access more documents
Once signed in, you may be able to access additional documents for your account.
EnviteC Cross Reference List | Tech Support
Resusable wrap pulse oximetry sensors. Easy to clean and flexible applications.
EnviteC by Honeywell
Por favor, faça login para acessar documentos adicionais.
Após o login, você terá acesso a documentos adicionais vinculados à sua conta.
Certificado
Others
Instructions for Use
Por favor faça login para ver os números de peças disponíveis para compra com base na sua conta Entrar
Filters
Por favor faça login para ver os números de peças disponíveis para compra com base na sua conta Entrar